The power of autogenous vaccines (autovaccines)
Vaccines are without doubt the most important pharmaceutical tool to prevent animal disease and improve animal health & welfare in livestock animals. Both registered and autogenous vaccines are an important player in this field.
Autogenous Vaccines: Bridging the Gaps in Disease Prevention
Vaccines are vital for animal health, yet registered vaccines may not cover every disease and animal species. This is where autogenous vaccines step in.
Autogenous vaccines are specifically produced for animals of a specific farm or animals that belong to the epidemiological unit of that specific farm. Autogenous vaccines are addressing the limitations of registered vaccines and enabling rapid response to emerging pathogens.
Addressing Societal Demands
With the pressure to reduce antimicrobial use and prioritize animal welfare, autogenous vaccines offer a preventive approach, aligning with societal demands.
At Dopharma, we offer services and support to our customers during the process of composing the vaccine to a follow-up on efficacy and safety of our Dophavacc vaccines.
Contact us to learn more about herd-specific disease-management and Dophavacc autogenous vaccines.
Introducing Dophavacc®
Customized solutions for animal health: Dophavacc is the brand name of Dopharma‘s autogenous vaccines produced by Ripac-Labor.
Ripac-Labor offers over two decades of expertise in veterinary diagnostics and autogenous vaccine production. Ripac-Labor is authorized for autogenous vaccine production by the German authorities, adheres to stringent regulatory standards and is an active member of EMAV.
The production of Dophavacc®
The Production Process: Efficacy
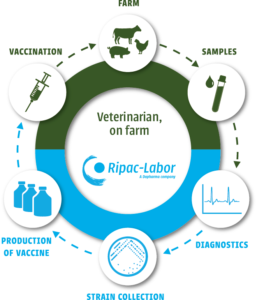
The basis of an effective autogenous vaccine lies in proper performed diagnostics. So, Dophavacc production begins with correct sampling on the farm followed by precise diagnostic investigations in the lab. After the isolation and identification, relevant pathogens are stored in RIPACs strain collection for a possible vaccine production.
Commitment to Quality Assurance
Ongoing investments in R&D and production facilities ensure quality and compliance, also with current and future regulatory requirements.
For consultation on diagnostics and/or the composition of the vaccine, please feel free to contact the local Dopharma technical support team.
About Dopharma
Dopharma is a family-owned veterinary pharmaceutical company based in the Netherlands. Since its establishment in 1969, Dopharma has been distributing high-quality veterinary medicines for livestock. Starting as a one-man operation, Dopharma has grown into a company with 9 production lines in Raamsdonksveer, the Netherlands. Dopharma’s products are distributed in over 70 countries through 10 local subsidiaries and approximately 60 distribution partners.
Dopharma is 100% dedicated to livestock animals. The broad product range today includes antimicrobial, antiparasitic and anti-inflammatory products, hormone preparations, nutritional supplements and autogenous vaccines.
Read more on www.dopharma.com
About RIPAC-LABOR
Ripac-Labor was founded in 2002 and started with a strong specialization in waterfowl and chicken. This is also reflected by the company name RIPAC, an acronym that stands for Riemerella, Pasteurella, Clostridia and Coli.
In 2007, the diagnostic laboratory and vaccine production moved to GO:IN at Potsdam Science Park in Golm, Germany. To be able to further expand the business and to be able to implement a quality management system, Ripac-Labor offered their majority shares to Dopharma in 2017. In 2021 Ripac-Labor became a 100% subsidiary of Dopharma International B.V. In the same year, Dopharma acquired land in Golm at the science park to develop and construct a new GMP compliant production facility.
Read more on www.dopharma-ripac.com

